February 26, 2026
3 min read
These cells in the body remember fat. Here’s what that means for weight loss
Obesity leaves a lasting imprint on fat and immune cells in ways that might make weight regain harder to avoid
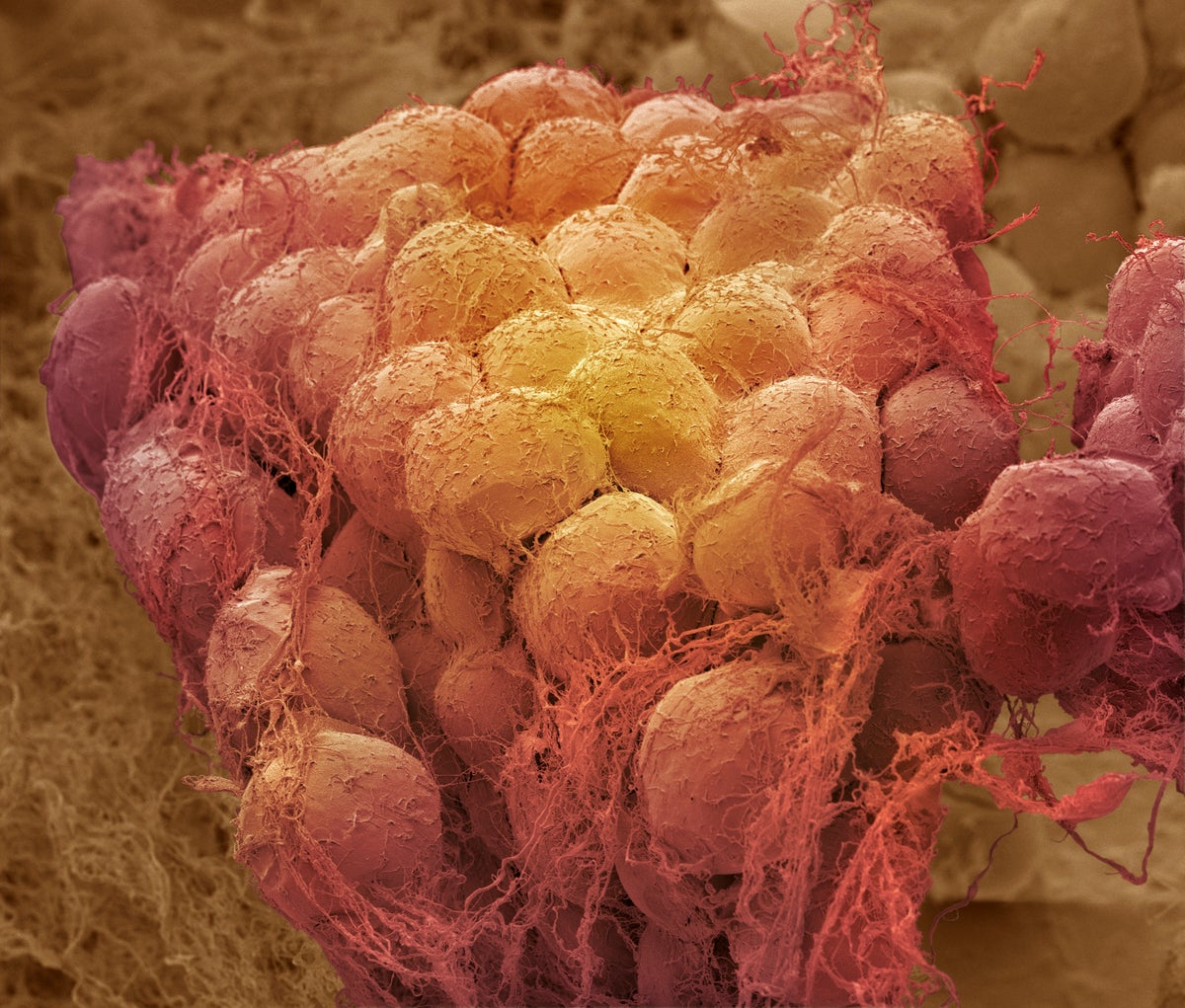
Colored scanning electron micrograph (SEM) of a sample of fat tissue, showing fat cells (adipocytes).
STEVE GSCHMEISSNER/SCIENCE PHOTO LIBRARY/Getty Images
Weight loss is notoriously hard to maintain. Within a few years most people regain the pounds they initially lose, whether through dieting, exercise, surgery or weight-loss medications such as the popular glucagonlike peptide 1 (GLP-1) drugs. Recent research suggests fat cells’ “memory” could explain why.
Cells that store fat, or adipocytes, and immune cells, such as macrophages, that live in fat tissue can remember weight long after it’s lost. And scientists suggest obesity causes lasting changes to those cells that make it easier to revert the body to a state of obesity, even after significant weight loss. The changes are etched into the cells’ epigenome, the instructions that tell each cell to read specific genes that control their function, explains Ferdinand von Meyenn, who studies nutrition and metabolic epigenetics at the Swiss Federal Institute of Technology Zurich. This ensures that a liver cell doesn’t suddenly behave like a neuron, for example. In people with obesity, lasting epigenetic changes might prime the body to regain weight more easily if they consume more calories.
Von Meyenn’s team measured gene activity in individual cells with a technique called RNA sequencing to compare fat tissue from people with obesity before they had bariatric surgery with similar tissue from people who didn’t have obesity. Even after the participants with obesity had lost about 25 percent of their body mass index after their surgery, some of their genes remained out of whack. This suggests some fat tissue didn’t recover from obesity—certain genes controlling metabolic function and inflammation remained abnormally switched on or off, for example, von Meyenn explains.
On supporting science journalism
If you’re enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Previous studies showed that fat cells in obese mice also retain epigenetic alterations even after the animals slim down. When they were later fed a high-fat diet, these mice regained weight faster than control animals. Lab tests showed fat cells from the obese mice absorbed glucose and lipids more readily. Fat cells normally take in sugars and lipids, von Meyenn says, but these obesity-altered cells appeared “slightly tweaked” to absorb more of those nutrients.
Other research groups have demonstrated that immune cells might also remember prior weight. When a person gains weight, different types of immune cells infiltrate expanding fat tissue, likely as a stress response, explains William Scott, an obesity researcher at Imperial College London. His research showed that, after bariatric surgery, the number of immune cells in people’s fat tissue decreased dramatically, but not everything completely reset. The immune cells retained the inflammatory characteristics that developed when the people had obesity.
Research in mice has echoed this finding; even after weight loss, macrophages retain epigenetic changes that keep inflammatory genes more active than usual. Another mouse study shows that weight cycling—losing and regaining weight—can intensify these immune cell changes and worsen metabolic health more than never losing the weight.
How long this epigenetic memory lasts isn’t clear, but fat cells can persist for up to a decade in humans, giving those cells the potential to maintain changes long-term, von Meyenn says. And fat may not be the only tissue involved. “I believe there are changes happening in the brain, in the liver, in the muscle,” says von Meyenn, who plans to investigate these areas next.
These findings don’t mean weight loss is futile; even short-term weight loss correlates with improvements in metabolic health. But the research might help explain why weight relapse is so common and why avoiding weight gain matters. In an environment and society that make weight gain prevalent, prevention is much easier said than done, von Meyenn says. Researchers, including von Meyenn, are now exploring whether fat cells can be treated to rewrite these epigenetic changes to make weight loss more sustainable and whether different types of weight-loss intervention have different effects on these cells.
“There’s a big drive to make our weight-loss drugs [such as GLP-1 medications] more potent” to cause greater weight reductions, Scott says, “but we really need to do better at maintaining weight loss once it’s happening.”
It’s Time to Stand Up for Science
If you enjoyed this article, I’d like to ask for your support. Scientific American has served as an advocate for science and industry for 180 years, and right now may be the most critical moment in that two-century history.
I’ve been a Scientific American subscriber since I was 12 years old, and it helped shape the way I look at the world. SciAm always educates and delights me, and inspires a sense of awe for our vast, beautiful universe. I hope it does that for you, too.
If you subscribe to Scientific American, you help ensure that our coverage is centered on meaningful research and discovery; that we have the resources to report on the decisions that threaten labs across the U.S.; and that we support both budding and working scientists at a time when the value of science itself too often goes unrecognized.
In return, you get essential news, captivating podcasts, brilliant infographics, can’t-miss newsletters, must-watch videos, challenging games, and the science world’s best writing and reporting. You can even gift someone a subscription.
There has never been a more important time for us to stand up and show why science matters. I hope you’ll support us in that mission.